Nuclear Process Heat for Industry
- Nuclear energy is an excellent source of process heat for various industrial applications including desalination, synthetic and unconventional oil production, oil refining, biomass-based ethanol production, and in the future: hydrogen production.
- For most major industrial heat applications, nuclear energy is the only credible non-carbon option.
- Light water reactors produce heat at relatively low temperatures in relation to many industrial needs, hence the technology focus has been on high-temperature gas-cooled reactors (HTR) and more recently on molten salt reactors (MSR) producing heat at over 700 °C.
- In 2019 there were 79 nuclear reactors used for desalination, district heating, or process heat, with 750 reactor-years of experience in these, mostly in Russia and Ukraine.
Two information papers in this series address the application of nuclear energy to desalination and hydrogen. This page covers other applications, but includes ethanol production.
Note that the unit MWt (megawatts thermal) is frequently used, with 3 MWt corresponding to about 1 MWe when electricity is generated.
The potential application of nuclear heat depends mainly on the temperature required. With reactor output temperatures of up to 700 °C there is a wide range of possible applications, at 900 °C there are further possibilities, and at 950 °C an important future application to hydrogen production opens up. About 20% of US energy consumption goes into process heat applications, compared with 35-40% into electricity. In this 20%, replacing fossil fuels with nuclear heat promises much in energy security, price stability and reduced regulatory risks, and it is the only option if carbon dioxide emissions are to be avoided.
High-temperature reactors for process heat
The most advanced high-temperature gas-cooled reactor (HTR) project is China's HTR-PM, based on its successful HTR-10 prototype. The demonstration unit being built in Shidaowan links twin 250 MWt units with 750 °C outlet temperature to a 210 MWe steam turbine.
The Nuclear Cogeneration Industrial Initiative (NC2I), part of the Sustainable Nuclear Energy Technology Platform (SNETP) in the European Union (EU), is focused on HTRs producing 550 °C steam for a variety of industrial applications. The Gemini+ project launched in September 2017 is an outcome of this, involving the EU and international partners (Japan, South Korea and the USA) and coordinated by the Polish National Centre for Nuclear Research (NCBJ). This project is funded under the Euratom programme and aims to provide a conceptual design of a high temperature nuclear cogeneration system that supplies process steam to industry, a licensing framework for this system and a business plan for a full-scale demonstration.
Poland has 13 large chemical plants which need 6500 MWt of heat at 400-550 °C. It is considering the potential role of HTRs for this, and very high-temperature gas-cooled reactors (VHTR) for hydrogen production. Deployment of HTRs for industrial heat production was included in the government’s July 2016 draft strategy for development. An early target is building a cogeneration HTR of 200-350 MWt for heat, and before this a 10 MWt experimental HTR at Swierk.
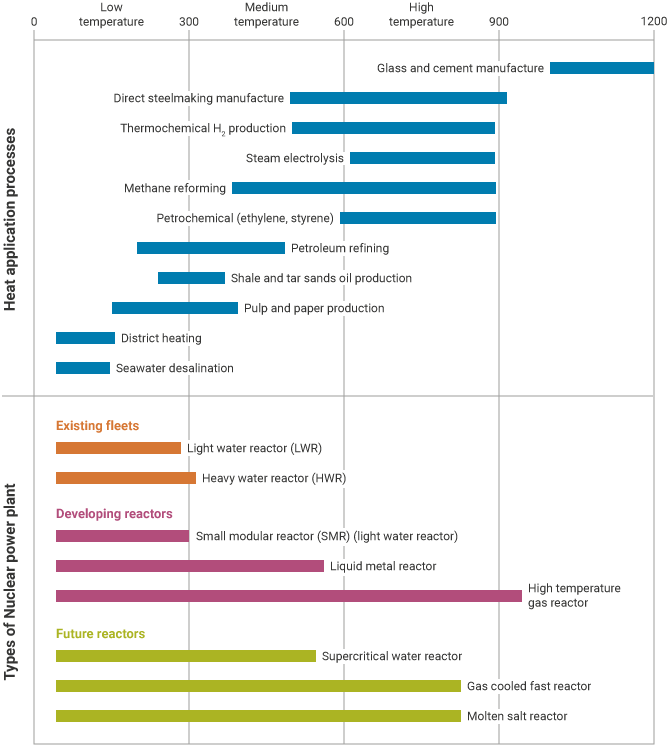
Temperature ranges of heat application processes and types of nuclear power plant (source: IAEA2)
In the USA the Department of Energy (DOE) since 2005 has had a programme (with diminishing funding) to develop a Next Generation Nuclear Plant (NGNP) which is to be an HTR capable of producing hydrogen, electricity or process heat. Initially this was focused on eventual hydrogen production, which requires very high temperatures with corresponding technological and cost challenges. Other process heat applications would broaden the end-user base for nuclear energy. The prospect of an operating prototype by 2021 receded. Industrial partners in the endeavour, with 11 member companies including reactor vendors Areva and Westinghouse, utility Entergy, and potential end-users of electricity and process heat such as Dow Chemical and ConocoPhillips, then set their sights on a more readily-achievable 750°C outlet temperature for process heat rather than hydrogen production particularly. The NGNP was to be built at Idaho National Laboratory (INL) with substantial cost share intended from industry.
Early in 2012 the NGNP Industry Alliance announced that it had selected the 625 MWt Areva HTR design, known as Antares, for further development. This would be run with helium as primary coolant at 750 °C and a two-loop secondary steam cycle.
More recently, the NGNP Industry Alliance has been collaborating with the NC2I on the design and regulatory framework for HTR technology and.in September 2017 launched the GEMINI+ project together with an international consortium to demonstrate high temperature nuclear cogeneration with HTR technology (see above). The NGNP Industry Alliance has evaluated US sites for a demonstration plant including the Piketon site in Portsmouth, Ohio; Odessa/Midland, Texas; a former Georgia Power coal power plant site; and Idaho Falls. All were found to be suitable but Piketon was judged best for electricity, process heat and hydrogen.
A 2011 evaluation by INL for a typical HTR multi-module plant showed that high-temperature process heat and electric power for use by energy-intensive industries can be produced at an energy price that is equivalent to natural gas in the $6-$9/GJ range. Areva's estimates support this.
In October 2015 a DOE assessment said: “The HTR, with outlet temperature limited to less than 800 °C, is suitable for near-term deployment as a demonstration reactor.” It noted US government and industry investment in HTRs since the 1960s, totalling over $3 billion.
In South Korea the Nuclear Hydrogen Development & Demonstration (NHDD) project is focused on HTR technology for both hydrogen production and liquid hydrocarbons. It was launched in 2006 as a key programme supported by the Ministry of Science and Technology (now the Ministry of Science and ICT). The Korean Atomic Energy Research Institute (KAERI) earlier submitted a Very High Temperature Reactor (VHTR) design to the Generation IV International Forum. The VHTR is to produce hydrogen on a large scale, with 300 MWt modules each producing 30,000 tonnes of hydrogen per year. KAERI had expected engineering design to be completed by 2014, construction start 2016 and operation of a demonstration reactor in 2020, with commercialisation about 2025. R&D focus is on development of corrosion-resistant high-temperature materials and components and refractory coated-particle fuel.
KAERI also has a research partnership with China's Tsinghua University focused on hydrogen production, based on China's HTR-10 reactor.
In Japan, JAEA has confirmed the safety of HTRs, and in April 2004 a coolant outlet temperature of 950 °C was achieved in its High-Temperature Engineering Test Reactor (HTTR) – a world first; and in 2009 it ran at 950 °C for 50 days, opening the way for direct thermochemical hydrogen production. In January 2019 it produced hydrogen using the iodine-sulfur process over 150 hours of continuous operation in its integrated process loop of 100 L/h design capacity, operating at 30 L/h. Meanwhile JAEA plans to make hydrogen by steam reforming natural gas, using HTTR’s high temperature.
Based on the HTTR, JAEA is developing the HTR50 nuclear steam supply system to operate at 750 °C and produce steam at 538 °C and 12.5 MPa. Thermal rating would be up to 200 MWt and it would have the same TRISO fuel as the HTTR. It involves Toshiba, Fuji Electric and KHI and a lead plant is planned for the 2020s.
Following this and moving to VHTR, JAEA plans the 600 MWt Gas Turbine High Temperature Reactor (GTHTR300) cogeneration system operating at 850-950°C to enable 300 MWe electricity generation using direct cycle gas turbine, with 370 MWt process heat for hydrogen production (120 t/day), steelmaking (650,000 t/yr) and other industrial applications including district heating and desalination (55,000 m3/day) using waste heat. The basic design has been completed, involving MHI, Fuji Electric, IHI, and NFI and the lead plant is expected in the 2030s. The IS hydrogen system beyond the intermediate heat exchanger would be non-nuclear grade, in line with other hydrogen production facilities.
In 2006 the South African PBMR board formalised the concept of a higher-temperature PBMR process heat plant (PHP) with reactor output temperature of 950 °C. The first plants were envisaged for 2016 and the applications would have been oil sands production and petrochemical industry (process steam), steam methane reforming for hydrogen and eventually thermochemical hydrogen production. Each 600 MWt module would produce about 200 tonnes of hydrogen per day, which is well matched to the scale of current industrial demand for hydrogen. In 2011 the programme was aborted due to lack of funds.
Process heat applications
| Process temperature | Up to 700 °C | Up to 900 °C | Up to 950 °C |
| Electricity production | Rankine (steam) cycle | Brayton (direct) cycle | |
|---|---|---|---|
| Utility applications | Desalination | H2 via steam reforming of methane or high-temperature electrolysis | Thermochemical H2 production |
| Oil and chemical industry | Tar/oil sands and heavy oil recovery, Syncrude, Refinery and petrochemical |
Syngas for ammonia and methanol | Thermochemical H2 production |
Recovery of oil from tar sands
From about 2003 various proposals have been made to use nuclear power to produce steam for extraction of oil from Alberta's northern tar sands deposits, as well as electricity for the major infrastructure involved.
About 1.05 GJ (28 m3) of natural gas is typically required to produce a barrel of bitumen by in situ methods (according to the Canadian Energy Resource Institute). This bitumen must then be upgraded to oil – natural gas is the raw material for hydrogen (by steam reforming) to break down the long-chain hydrocarbons to yield synthetic crude oil. The extraction overall represents a total energy intensity of 1.37 GJ per barrel of oil (each embodying 6 GJ), and in 2014 it accounted for one-third of Alberta's primary energy demand.
With production of 2.4 million barrels per day of crude bitumen in 2016 (half mined and half in situ), a great deal of gas is used and the cost exposure is considerable. Production is expected to reach 3.6 million barrels per day in 2026. In fact, Canadian natural gas is inadequate to supply the anticipated expansion in oil sands output and its use has major CO2 implications which are creating public concern – over 80 kg of CO2 per barrel is released in its production. Oil sands production accounts for 8.5% of Canada’s greenhouse gas emissions.
An intrinsic problem is that the steam supply needs to be semi portable as tar sand extraction proceeds, so relatively small reactors which could be moved every decade or so may be needed. One problem related to the provision of steam for mining is that a nuclear plant is a long-life fixture, and mining of tar sands proceeds across the landscape, giving rise to very long steam transmission lines and consequent loss of efficiency.
One proposal from Energy Alberta Corp. suggested that a single CANDU 6 reactor (about 1800 MWt) configured to produce 75% steam and 25% electricity would replace 6 million cubic metres per day of natural gas and support production of 175-200,000 barrels per day of oil. It would also save the emission of 3.3 million tonnes of CO2 per year. Other figures from Pebble Bed Modular Reactor (PBMR) proponents confirm that each 100 MWt will enable production of 10,000 barrels per day.
The Canadian Energy Research Institute (CERI) published a report in February 2009 saying that employing nuclear energy with (so far untested) carbon capture and storage in tar sands extraction and processing could make oil from that source cleaner than conventional oil with respect to its greenhouse gas and other emissions. The CERI report looked at both very large (1600 MWe) and multiple very small (10 MWe) nuclear reactors.
A more recent proposal is from Canada’s Terrestrial Energy, putting forward its Integral Molten Salt Reactor (IMSR), which the company aims to commercialise by 2021. It is considering 300 MWt units, producing simply steam.
The main difference between natural gas and nuclear steam generation is that a fuel-intensive process is replaced by a capital-intensive one, with no carbon emissions.
Oil refining
As well as separating the different components of crude oil by two distillation processes, an oil refinery typically breaks down the residual heavy or long-chain hydrocarbons in a catalytic cracker by adding hydrogen. The hydrogen is produced from natural gas, and it breaks down the long-chain hydrocarbons to yield synthetic crude oil (about 5 kg is used per barrel). This hydrogenation of heavy crude oil is a major use of hydrogen today. Overall, about 15-20% of the energy value of the crude oil is used in producing refined products.
Hydrogen production is by steam reforming of the natural gas, and gives rise to a lot of carbon dioxide. Nuclear power could make steam and electricity and use some of the electricity for high-temperature electrolysis for hydrogen production. (Heavy water and oxygen could be valuable by-products of electrolysis). Oil refineries account for about one quarter of the world's hydrogen demand.
Coal to liquids (CTL)
Various processes are used. Indirect liquefaction accounts for most world production, about 260,000 barrels/41,000 m3 per day. Coal is gasified to syngas and the product synthesized either to diesel and jet fuel, or to gasoline (Mobil or methanol MTG process). Direct liquefaction processes miss the syngas step and are either hydrogenation or pyrolysis and carbonization.
The Fischer-Tropsch (F-T) process was originally developed in Germany in the 1920s, and provided much of the fuel for Germany during the Second World War. It then became the basis for much oil production in South Africa by Sasol, which now supplies most of that country's diesel fuel. However, it is a significant user of hydrogen, catalyzing a reaction with carbon monoxide. The hydrogen is now produced with the CO by coal gasification, part of the gas stream undergoing the water shift reaction.
* Coal gasification produces both CO and H2 feed for F-T, and the water shift reaction turns some CO into CO2 and more H2, both stages using water. In F-T itself hydrogen plus CO converts to alkane hydrocarbons using a catalyst.
At Erdos, Inner Mongolia, China, Shenhua Coal Liquefaction Corp produces 20,000 barrels/3000 tonnes of oil per day from nearly 10,000 tonnes of coal from the Shenfu-Dongsheng coalfield, using US technology. The $2.06 billion Erdos CTL plant was commissioned in 2010 and in 2013 it produced 866,000 tonnes of oil products. It has total design capacity of 1.08 million tonnes, comprising diesel (621,000 t/yr), naptha (321,000 t/yr) and LPG (70,000 t/yr). Water supply is a constraint on operation, and it requires 7-12 tonnes of fresh water to produce one tonne of product, depending on whether direct liquefaction or indirect (based on F-T), along with 9 tonnes of CO2 and 4.8 tonnes of contaminated waste water according to critics. It uses coal hydrogenation at high temperature and pressure with a catalyst.
Shenhua proposes $7 billion plant in Ningdong, Ningxia, China – the Shenhua CTL plant to produce 80,000 barrels/ 13,000 m3/day, using Sasol’s indirect liquefaction technology, based on F-T. Sasol has withdrawn from the venture.
The hydrogenation (Bergius) process requires hydrogen to react directly with coal at high temperature and pressure. Pyrolysis and carbonization processes give less yield, and that usually needs refining. They are not commercial for CTL.
A nuclear source of hydrogen coupled with nuclear process heat would more than double the amount of liquid hydrocarbons from the coal and eliminate most CO2 emissions from the process.
Using simply black coal in the Sasol-type plant, 14,600 tonnes produces 25,000 barrels of synfuel 'oil' (with 25,000 tonnes of CO2).
A hybrid system uses nuclear electricity to electrolyse water for the hydrogen. Some 4400 tonnes of coal is gasified using oxygen from the electrolysis to produce carbon monoxide which is fed to the Fischer-Tropsch plant with the hydrogen to produce 25,000 barrels of synfuel "oil". Very little CO2 results, and this is recycled to the gasifier.
Biomass-based ethanol production
Commercial-scale ethanol production from cellulose in a biorefinery requires energy input. Two processes can be used: biochemical fermentation, or thermochemical conversion. Either way, the energy input needed to produce ethanol from cellulose materials (such as wood) is equivalent to 25 to 38% of the output, mostly as steam (though the thermochemical process also requires high temperatures).
As well as burning the lignin content of feedstock, some energy can readily be supplied by nuclear plants as cogeneration, on a carbon-free basis. In future, the lignin may be converted to liquid fuels, probably by hydrogenation. In this case, the yield of liquid fuels from biomass can be increased by about half, per unit of input. Much of the nuclear energy input is as low-temperature steam, essentially a by-product of electricity generation, and hence low-cost.
District heating
Many reactors in cold climates provide hot water for district heating, without significant penalty in electrical performance. This usually replaces fossil fuel sources, with significant reduction in carbon dioxide emissions. Most of the nuclear district heating has been developed within northern European or former Soviet states as the cold climate and long heating periods create favourable conditions for district heating and cogeneration development.
Apart from new reactors designed specifically for district heat, this service may be from a power reactor’s condenser circuit or from tapping into the secondary steam circuit. This is being demonstrated at the Haiyang nuclear power plant in China, to heat the whole city with 300-400 TJ/yr. It is planned for the new CAP1400 plant in Shandong, and for plants at Qinshan, Tianwan and Hongyanhe.
If simply tapping the condenser circuit as more widely done, water at around 100 °C at low pressure is required.
China has three low-temperature reactor designs specifically for district heating rather than power. CGN has the NHR-200 (200 MWt) at Daqing city, CNNC has the DHR-400 Yanlong (400 MWt), and SPIC has the 200 MWt LandStar-I which delivers hot water at 110°C with convection circulation through a heat exchanger. Ten units are planned in northeastern China, with the first under construction in Jiamusi and Daqing in Heilongjiang province. The two units at Jiamusi will provide steam to a biomass plant in summer.
Russia’s low-temperature reactor design specifically for district heating rather than power is the AST-500, proposed for several sites (500 MWt). The 2 x KLT40 floating nuclear power plant at Pevek also produces district heat.
Notes & references
References
1. International Energy Agency, The Future of Hydrogen – Seizing today's opportunities, Report prepared by the IEA for the G20, Japan (June 2019) [Back]
2. International Atomic Energy Agency, Opportunities for Cogeneration with Nuclear Energy (May 2017) [Back]
General references
Schultz, K.R. 2004, Use of the modular helium reactor for hydrogen production, Nuclear Engineer 45,2
Walters, Leon et al 2002, Transition to a nuclear/hydrogen energy system, The Nuclear Engineer 43,6
Shiozawa, S. et al 2003, Status of the Japanese development study of hydrogen production system using HTGR, KAIF/KNS conference
Schultz, K. et al, 2005, The Hydrogen reaction, Nuclear Engineering International, July 2005
Sakaba, N. et al, 2005, JAERI's Hot Stuff, Nuclear Engineering International, July 2005
Brown, Russell 2006, Critical Paths to a Post-Petroleum Age (ANL paper).
Barre, B. & Baquis, P-R. 2007, Oil and Nuclear Energy: Toward a Long-term Symbiosis?, WNA Symposium
Forsberg, C.W. 2008, Sustainability by combining nuclear, fossil and renewable energy sources, Progress in Nuclear Energy
Greene S.R. et al 2009, Integration of biorefineries and nuclear cogeneration power plants – a preliminary analysis, ORNL/TM-2008/102
Erdos Shenhua project: Yuzhuo Zhang, Shenhua Group Corporation Ltd., Shenhua Coal Conversion Technology and Industry Development (2007)
Methanol Institute
Carlos A. Murillo, Canadian Energy Research Institute, Oil Sands Industry Energy Requirements and Greenhouse Gas (GHG) Emissions Outlook (2015-2050) (August 2015)
Roger Arnold, The lowdown on hydrogen – part 2: production, Energy Post (21 April 2017)
Tim Wogan, Zeolite catalysts convert carbon dioxide to fuel, Chemistry World (13 June 2017)
Donald Hoffman, EXCEL Services, Nuclear High Temperature Heat for Industrial Processes – HTGRs, GEMINI, the PRIME Concept and the U.S. Advanced Reactor Effort, presented at a side event to the International Atomic Energy Agency 61st General Conference held in Vienna, Austria (18-22 September 2017)
International Atomic Energy Agency webpage on non-electric applications of nuclear heat
International Energy Agency, The Future of Hydrogen – Seizing today's opportunities, Report prepared by the IEA for the G20, Japan (June 2019)
The Royal Society, Nuclear Cogeneration: civil nuclear energy in a low-carbon future (October 2020)
International Atomic Energy Agency, Opportunities for Cogeneration with Nuclear Energy (May 2017)
Related information
Advanced Nuclear Power ReactorsDesalination
Hydrogen Production and Uses